Hemostasis and Sealing
Simple. Proven.
mediCipio A is a two-layer medical device designed for secure sealing
and hemostasis during invasive medical procedures.
Technology and Performance
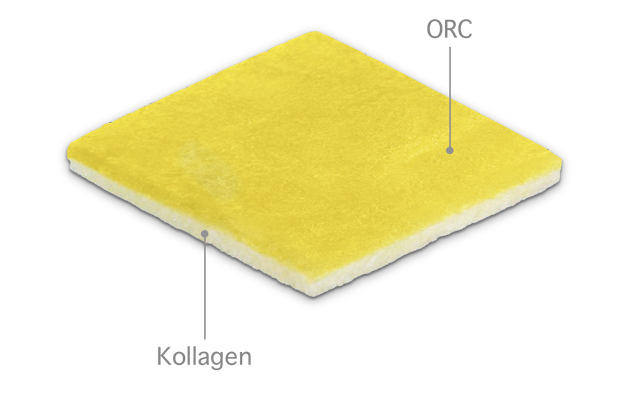
The medical device mediCipio A is designed for superior sealing and hemostasis in surgical procedures. It consists of oxidized regenerated cellulose (ORC) and porcine collagen, without the use of human fibrinogen or thrombin.
The dual-layer structure of the mediCipio A amtrix combines the reliable adhesion, hemostatic effect, and absorbency of the ORC layer with the stability, sealing capacity, biocompatibility, and wound-healing properties of the collagen layer.
We achieve these exceptional material properties through our proprietary manufacturing process, which incorporates patent-protected and other exclusive, meticulously balanced process steps.
Clinical Application
The soft patch is removed from the packaging and applied directly with the yellow side (ORC) onto the moist or rinsed tissue. No hydration or other preparation is required. Once pressed into place, it adheres immediately to provide sealing or hemostatic effects, depending on the application. The patch can be applied with overlapping layers; it remains in the body and is fully resorbed within weeks.

- Ease of use
- Surgical advantages
- Less bureaucracy and risk
Ready-to-use: No pre-moistening of the product.
Superior Handling: mediCipio A adheres to the wound, not to gloves or instruments—no prior cleaning of tools necessary.
Maximum Flexibility: The soft matrix offers easy and flexible adaptation to the wound site.
Customizable: Can be easily trimmed to size as needed.
Immediate Results: Instant adhesion after applying gentle pressure.
Non-swelling: No expansion after application, allowing for safe use even in close proximity to sensitive structures (e.g., nerves or brain tissue).
Anatomical Adaptation: Soft material allows for precise contouring to complex structures—such as over the cranial edge—for an even more effective seal.
Structural Integrity: The material remains intact during handling and application; no components or particles shed or detach from the matrix.
Suture-ready: Due to its high mechanical stability, the patch can be securely sutured (e.g., to the dura mater) without tearing.
Superior Absorption: Dry application enables higher fluid uptake compared to pre-moistened alternatives.
Rollable: Can be rolled for easy delivery, e.g. through trocars or in narrow approaches.
Pathogen Safety: Contains no human blood components, eliminating associated infection risks.
Simplified Consent: Patient disclosure regarding infection risks from blood-derived components is not required.
Reduced Administrative Effort: No documentation requirements under the Transfusion Act, as the product is free of blood-derived ingredients.
Compatibility with Autotransfusion: Can be used alongside cell salvage systems (Cell Saver), as it contains no thrombin or fibrinogen.
Extended Shelf Life: Storage 5 years at room temperature.
Superior Material Properties for Advanced Handling
mediCipio A is engineered for exceptional structural integrity while remaining remarkably soft and comfortable to handle. This high-quality, flexible matrix can be effortlessly trimmed to size without crumbling or shedding particles. Its mechanical stability ensures that the patch can be securely sutured—for example, to the dura mater—without tearing.
The material is easy to roll or shape, making it ideal for use through a trocar or surgical approaches in tight spaces. This flexibility of the soft material also allows for precise contouring to complex anatomical structures, such as over the cranial edge, for an even more effective seal.
Originally approximately 5 mm thick, the dry matrix is applied directly to the moist area where hemostasis or sealing is required. By applying gentle pressure—for example, using a moist compress—it adheres immediately to the tissue to create a thin, protective layer. The non-swelling nature of the material allows for safe use near sensitive structures.
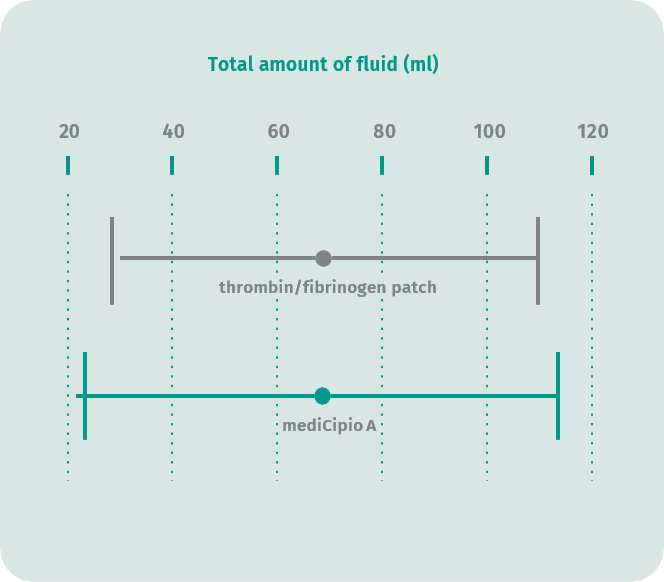
Primary endpoint: Total fluid volume at the time of removal of the Redon drainage system following thyroidectomy
Clinical evidence
In a prospective, randomized clinical trial with 150 patients, it was confirmed that mediCipio A is as effective in inducing hemostasis as a leading thrombin-fibrin matrix. Thyroid surgery was selected for this study because this type of procedure allows for easy measurement of fluid volume using a Redon drainage system.
Effective hemostasis was also confirmed when antiplatelet aggregation inhibitors were used.
Published in the Journal of Visceral Surgery in 2020, https://doi.org/10.1016/j.jviscsurg.2020.06.015.
Clinical Evidence in Neurosurgery
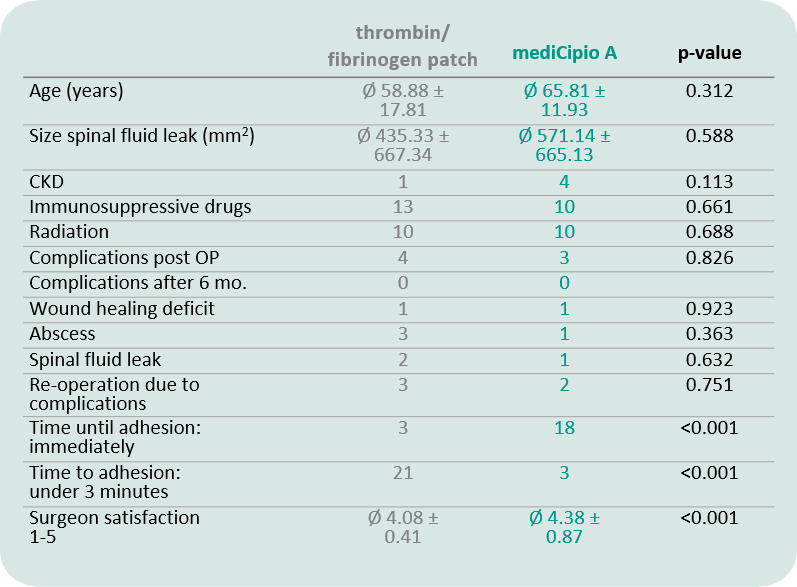
In a prospective clinical trial with a total of 45 neurosurgical patients, it was confirmed that mediCipio A is non-inferior to a leading thrombin-fibrin matriregarding postoperative complication rates, including CSF leakage, infections, and the need for re-surgery.
Surgeon satisfaction was significantly higher in the mediCipio A group, and the time to adhesion was significantly shorter.
Key facts about the study:
- Conducted by: Bayreuth Hospital under the direction of Prof. Dr. Stefan Linsler.
- Status: The study was continued and has been expanded to include 110 patients.
- Publication: The full study will be published in 2026. The preliminary results shown here were presented at EANS 2025 in Vienna in October 2025: https://www.sciencedirect.com/science/article/pii/S2772529425013694

Available sizes
Downloads
IFU mediCipio A
118 kB
Declaration of Conformity mediCipio A
88 kB
Surgery
mediCipio A is used for hemostasis and tissue sealing in surgical procedures. It is completely absorbable and consists of oxidized regenerated cellulose (ORC) and bio-porcine collagen—without the use of human blood components or synthetic materials.
Wound healing
mediCipio C is a fully absorbable wound-healing product made from pure bio-porcine collagen; its special pore structure helps absorb exudate and maintains a moist environment that promotes wound healing.
Oral Surgery
mediRegis C is a fully resorbable bio-porcine collagen membrane for dental use that promotes hemostasis as well as bone and tissue regeneration and can be safely used even in patients with coagulation disorders.